Cancer represents one of the most complex diseases in the world. The vast number of cancer types that vary in pathology and response to treatment continue to pose a problem. CAR-T and TCR therapies represent the next step in developing effective immunotherapy to address these challenges. The latest innovations in these methods offer hope for treatment-resistance patients and potentially rare cancer types.
Introduction to cellular immunotherapy
Adoptive T cell (ACT) therapy is a type of cancer immunotherapy which uses the specificity of T cells to selectively target tumour antigens. Initially, tumour-specific T samples are harvested from resected tumours, to which the sample is expanded via in-vitro methods. This method was relatively limited, as not all tumours can be removed depending on location.
This meant that not all patients were eligible for this type of therapy. However, expansions in genetic engineering technology have seen ACT develop into an option for a multitude of cancer types. The two main ACT methods under development in oncology are chimeric antigen receptor (CAR-T) therapy and T cell receptor (TCR) therapy.
CAR-T
CAR-T cell therapy allows the production of T cells that can target almost any naturally occurring protein structure on any cell. T cells are engineered to express a chimeric antigen receptor (CAR), a type of receptor protein that can recognise and bind to specific antigens on cancer cells.
To avoid damaging healthy tissue which could cause detrimental consequences, the specificity of the target tumour antigens is crucial to ensure T cell-driven mechanisms occur only in the cancer cells. Viral-based vectors are the main method of producing CAR-T cells in the transferring of genetic information. Lentiviral vectors are a popular choice due to their ability to “target nondividing cells with relative ease, whereas in gamma retroviral vectors, the transduction rates into nondividing cells are significantly lower.” Non-viral vectors are emerging as a popular alternative to viral vectors, which continue to pose a risk for oncogenic mutation.
The current alternative is the sleeping beauty (SB) transposon system. Summarised in a 2020 paper, the system is a “nonviral DNA transfer tool capable of efficiently mediating transposition‐based, stable integration of DNA sequences of choice into eukaryotic genomes.” The stable integration of genetic material is one of the main strengths to this approach of producing CAR-T cells. This reduces the chance of genetic changes, which can cause mutations and compromise patient health.
TCR Therapy
The TCR is expressed by all T cells. Its role is to recognise antigen structures. In comparison to CARs, which rely on recognition of antigens, TCRs can detect a broader range of targets, including neoantigens and viral oncoproteins. TCR is another form of ACT, but with the expression of native or transgenic αβ-T cell receptors. ‘Native’ simply refers to genetic material from the original organism, while ‘transgenic’ means the artificial introduction of DNA from another species.
Clinical success
CAR-T therapy has so far shown great success in clinical research. A 2017 study investigating the efficacy of Yescarta, a CAR-T cell therapy, found that the “immunotherapy slowed or stopped the growth of cancer in 82% of patients, and more than half (54%) of the cancers disappeared completely”. The patient group had previously shown resistance to treatment. Significantly, this demonstrates a potential alternative treatment option for those who have shown a poor response to treatment like chemotherapy. Furthermore, the efficacy of this immunotherapy in the long term was further supported by the same study, which saw that “nearly 15.4 months later, approximately 40% of patients still showed no signs of cancer”.
TCR therapy has existed since the early 2000s. It was not until 2017, however, that the first clinical trial using a WT1-specific TCR-T therapy occurred.
WT1 is an antigen associated with tumours, expressed in acute leukaemia and myelodysplastic syndrome (MDS). Of the eight patients with refractory acute myeloblastic leukaemia, “two showed a transient decrease in blast counts in bone marrow, which was associated with haematopoiesis recovery”. In the same study, “four out of five patients who had persistent T cells at the end of the study survived longer than 12 months”. The significance of this is an obvious improvement in the prognosis for patients with refractory leukaemia, which is notoriously difficult to treat. Furthermore, it shows the potential to engineer TCRs relative to the cancer type, which offers hope for a multitude of patients with rare cancer.
Safety concerns
Both methods of immunotherapy pose potential patient safety issues. Cytokine release syndrome (CRS) can occur after CAR-T therapy as a result of the elevated levels of inflammatory molecules by the CAR-T cells. CRS is a common condition that can develop as a result of immunotherapy, causing long-term fever, hypotension and organ problems. The most severe of cases can require intensive care, including the use of ventilators. CRS is a common consequence of CAR-T therapy, however “studies have shown that patients with massive lesions, patients with a high tumor burden, patients with complications, and patients with CRS after 3 days of CAR-T cell infusion are prone to severe CRS”.
A major obstacle for TCR therapy is on-target, off-tumour toxicity. Despite showing a high affinity for target tumour antigen, the selectivity of TCR is poor, which together can cause issues in healthy tissue with similar antigen expression. An example of this is the toxic effect that can arise in melanocyte-rich tissues. Melanoma is a form of cancer which begins in the melanocytes. Unfortunately in T cells expressing a TCR for the melanocyte differentiation antigen MART-1, the poor selectivity results in healthy tissue becoming targeted, which in one case resulted in a fatal adverse event.
CAR-T and TCR therapy are prime examples of innovative, targeted drug therapies, which overcome the poor selectivity of systemic chemotherapy. With continuous research to address safety issues, this is an important step in precision cancer treatment with fewer side effects and potentially a better clinical response.
Charlotte Di Salvo, Lead Medical Writer
PharmaFeatures
Subscribe
to get our
LATEST NEWS
Related Posts

Immunology & Oncology
The Silent Guardian: How GAS1 Shapes the Landscape of Metastatic Melanoma
GAS1’s discovery represents a beacon of hope in the fight against metastatic disease.
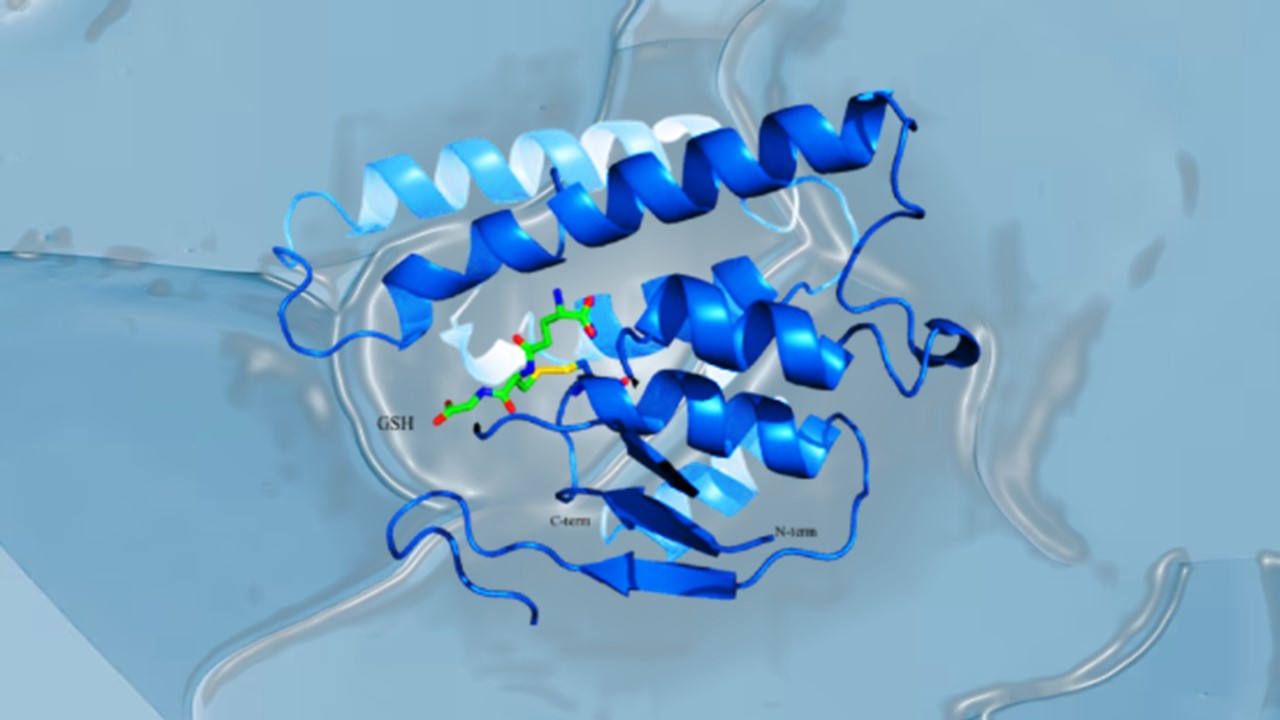
Immunology & Oncology
Resistance Mechanisms Unveiled: The Role of Glutathione S-Transferase in Cancer Therapy Failures
Understanding this dual role of GSTs as both protectors and accomplices to malignancies is central to tackling drug resistance.
Read More Articles
Myosin’s Molecular Toggle: How Dimerization of the Globular Tail Domain Controls the Motor Function of Myo5a
Myo5a exists in either an inhibited, triangulated rest or an extended, motile activation, each conformation dictated by the interplay between the GTD and its surroundings.
Designing Better Sugar Stoppers: Engineering Selective α-Glucosidase Inhibitors via Fragment-Based Dynamic Chemistry
One of the most pressing challenges in anti-diabetic therapy is reducing the unpleasant and often debilitating gastrointestinal side effects that accompany α-amylase inhibition.