About the Interviewee

Aditya Tallapragada is the President of AKT Health, Inc and Interakt, Inc.
Dr. Aditya Tallapragada is a healthcare and technology leader with over a decade of experience spanning life sciences consulting, clinical research, enterprise blockchain, and artificial intelligence. As President of AKT Health Inc. and Interakt Inc., and Co-Founder & CTO of TAMP, he has guided biopharma, healthcare, and technology clients in adopting data-driven digital transformation strategies that enhance clinical, commercial, and medical operations while advancing customer engagement. He also serves as Chief Impakt Officer at Medifakt, where he combines strategic consulting with expertise in health economics, market access, and digital health innovation.
Dr. Tallapragada holds a Doctor of Business Administration in Leadership and Management from the European Institute of Management & Technology, an MBA in Finance from NIBM Chennai, and a B.Tech in Mechanical Engineering from Jawaharlal Nehru Technological University. He further specialized in pharmacoeconomics and outcomes research through certifications from the International Society of Pharmacoeconomics and Outcomes Research (ISPOR), reinforcing his dual expertise in healthcare strategy and quantitative analysis. His early career as a Quantitative Analyst in the financial sector honed his skills in econometric modeling and market analytics before transitioning fully into healthcare and technology leadership.
Throughout his career, Dr. Tallapragada has held senior roles at MakroCare and Truven Health Analytics (IBM Watson Health), where he managed clinical development, regulatory affairs, and health economics projects for global pharmaceutical clients. His work included KOL mapping, HEOR models, oncology Phase II and III clinical studies, and regulatory strategy for product launches across Asia and international markets. At Truven, he led projects in global dossier development, stakeholder management, and value demonstration packages that supported major market access and reimbursement pathways.
Today, Dr. Tallapragada is recognized for his ability to integrate healthcare and frontier technologies. Through Interakt, he advances decentralized applications for healthcare, real estate, and supply chain industries leveraging blockchain; through TAMP, he drives physician digital upskilling programs reaching over 90,000 doctors across medical associations; and through AKT Health, he positions biopharma clients at the forefront of digital excellence with tailored clinical, commercial, and medical strategies. His career reflects a rare combination of clinical development expertise, financial acumen, and digital innovation leadership, making him a sought-after advisor and strategic partner across global healthcare and technology ecosystems.
The Discussion
Leadership Origins: From Engineering to Executive Vision
[Engr. Dex Marco]: It’s such a pleasure to have you here with us today, Dr. Tallapragada. So Aditya, your career has been
shaped by a unique confluence of mechanical engineering, business strategy, and advanced management
research. Can you share the key milestones and mindset shifts that led to your appointment as President of
AKT Health, Inc., and how your multidisciplinary background has informed your leadership style?
[Dr. Aditya]: Thank you for having me, Dex. If I had to describe my professional journey, I’d say it’s been a continuous evolution through systems, financial, healthcare, and now intelligent clinical ecosystems.
I began as a quantitative analyst in finance, building pricing models and decoding patterns in complex datasets. It was intellectually rewarding, but it left me wondering: was I solving problems that truly mattered? That question led me to pivot into healthcare.
I started with business development and regulatory support in Japan, working closely with pharma clients, sponsors and CROs across East Asia. It gave me a ground-level view of how fragmented, paper-heavy, and siloed clinical operations still were. I remember thinking: this system isn’t just inefficient; it’s unfair to the patients and practitioners trying to navigate it.
Working in APAC-based clinical research environments gave me hands-on insight into how trials were conducted, from regulatory strategy to operational execution and digital enablement. It sharpened my understanding of the sponsor pain points and opportunities for scalable technology intervention.
Later, at IBM Watson Health role, I focused on health economics, outcomes research, and stakeholder mapping. I wasn’t designing solutions yet, but I was engaging with some of the toughest strategic questions around value demonstration, market access, and real-world evidence. That experience taught me and led to a shift in mindset, from solving static problems to reimagining dynamic, system-level solutions. I realized the same pattern recognition I had developed in finance could be applied to predicting trial risk, site performance, or even patient dropout.
That intersection where precision analytics meets human impact became my North Star. Founding AKT Health was a natural step forward. I didn’t want just to consult, I wanted to build. Today, my engineering background ensures we create scalable, modular systems; my experience in APAC clinical research ensures those systems are aligned with real-world workflows; And my passion for human-centered innovation ensures our tools serve patients, not just protocols.
We call it empathetic engineering technology that supports patients, sites, and sponsors alike. And in many ways, I believe our most impactful work is still ahead of us.
Predictive Intelligence: Translating Complexity into Clarity through Hakase AI
[Dex]: Managing clinical complexity demands advanced analytical frameworks and decision-making tools. Can you elaborate on how Hakase AI is being leveraged to transform multidimensional clinical data into predictive intelligence that accelerates risk mitigation and enhances protocol adherence?
[Dr. Aditya]: Absolutely. Hakase AI is the intelligence engine that underpins our entire digital health ecosystem.
Hakase in Japanese means a master of knowledge, like an “expert” or “PhD-level mastery”. And that’s fitting, because it functions like a hyper-specialized clinical scientist, operating 24/7 with perfect memory and pattern recognition.
That’s the ethos behind Hakase AI; it’s a purpose-built intelligence platform designed specifically forlife sciences, spanning three integrated domains: Clinical research, Commercial strategy, and Patient care. Unlike generic AI wrappers, Hakase combines domain-trained biomedical models with 60+ pre-integrated healthcare datasets. The result is an ecosystem that accelerates drug development, optimizes commercial decision-making, and personalizes patient care:
(1) In Clinical research, Hakase predicts trial success, optimizes protocols, automates CDISC-compliant submissions, and reduces queries by up to 78%.
(2) In the Commercial domain, it provides biotech-investor matching, pricing optimization, and real-time market access intelligence, helping companies make investment and launch decisions 3x faster.
(3) In Patient care, it generates personalized treatment plans, provides predictive alerts for early intervention, and supports coordinated care, improving patient retention by 85% and reducing readmissions by 20%.
Focusing on the Clinical module, Hakase AI acts like a hyper-specialized scientist working 24/7 with perfect recall and predictive reasoning. The challenge in modern clinical trials isn’t just data volume; it’s dimensionality, fragmentation, and the need for real-time interpretation. Hakase ingests structured and unstructured data; protocols, wearable streams, EMRs, eCOAs, site logs, lab data, even public datasets, and applies domain-trained AI models to deliver actionable intelligence.
Some examples:
(1) Protocol Optimization: Hakase reduces protocol amendments by 50% through predictive modeling.
(2) Site Performance Prediction: With 92% accuracy, it identifies high-performing sites, improving trial outcomes.
(3) Patient Recruitment: AI-powered matching algorithms accelerate enrollment by 42%.
(4) Document Intelligence: NLP models review multilingual trial documents, flagging protocol deviations in real-time.
(5) Predictive Modeling: It links signals across data streams, such as wearable data, ePRO entries, or lab results to proactively detect risks and improve adherence.
But what makes Hakase more than just a predictive engine is its ability to act proactively. It doesn’t just identify risks, it orchestrates the response. If dropout risk rises, Hakase triggers timely nudges through our Pulse engagement platform. If safety signals emerge, it escalates alerts based on severity and confidence scores. Most importantly, it learns continuously. Every prediction, every outcome, and every intervention feedback loop helps refine the models. With every study, its accuracy improves, and the interventions become more targeted and effective.
So when we talk about Hakase AI, it’s not a static analytics tool. It’s a living intelligence ecosystem that learns, adapts, and orchestrates responses across clinical, commercial, and care domains. In trials, that means shifting from reactive firefighting to proactive, adaptive design, helping sponsors, sites, and patients navigate complexity with clarity.
And unlike black-box AI, Hakase is fully auditable. Every decision it makes, from risk predictions to protocol adjustments, is logged, traceable, and explainable, which is vital for regulatory acceptance.
So in short, Hakase AI is not a siloed feature; it’s the cognitive fabric that connects every layer of our platform. It’s transforming the way we conduct trials from being reactive to becoming truly anticipatory.
Modular Clinical Trial Suites: Engineering Flexibility into Clinical Systems
[Dex]: AKT Health, Inc. has made significant strides in developing modular and interoperable trial solutions that address the evolving needs of sponsors and CROs. How are you approaching the design and integration of modular clinical trial suites to ensure interoperability, scalability, and regulatory alignment across diverse trial environments?
[Dr. Aditya]: This aligns closely with how we’ve engineered our clinical infrastructure from the ground up.
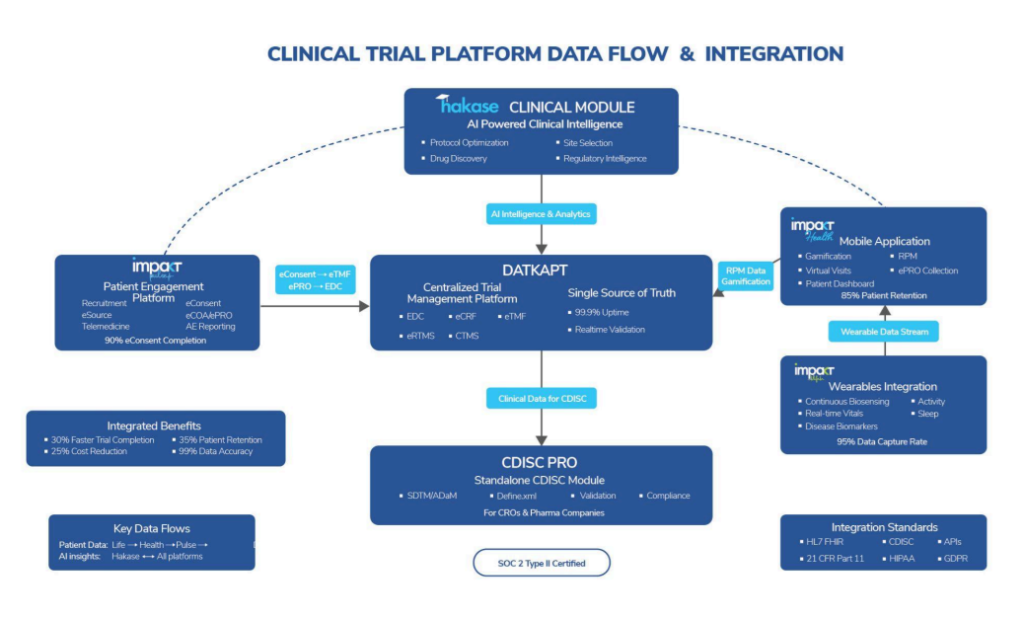
Traditional clinical tech stacks tend to be monolithic, rigid, hard to scale, and prone to failure when one component falters. We’ve taken a different approach. The Impakt Clinical Suite is built around what I call “intelligent modularity”, independent platforms that deliver value individually, but create exponential impact when combined.
Let me walk you through the four core modules and how we approach integration:
(1) Datakapt acts as the foundational clinical trial engine. It delivers enhanced data accuracy and operational visibility across EDC, eCRF, eTMF, RTMS, and CTMS components. Built on HL7 FHIR and CDISC-compliant APIs, it seamlessly plugs into sponsor ecosystems or operates standalone. It’s not just interoperable, it’s infrastructure-agnostic.
(2) Pulse powers patient and site engagement. Its modularity means you can deploy just the eConsent functionality for an early-phase trial or leverage the full suite, including ePRO, telehealth, and site dashboards, for complex decentralized studies. We’ve designed it to reduce time-to-recruitment and improve retention without overwhelming users.
(3) Health is our SaMD layer for patient-facing interaction. What sets it apart is its adaptability. The same engine can serve as a lightweight symptom tracker in one trial, and a multilingual lifestyle management app in another all while maintaining uniform backend logic and data standards.
(4) Impakt Life integrates a full suite of medical-grade wearables: ECG patches, smart rings, BP cuffs, and CGMs, built for continuous monitoring. While each device can function independently, the value multiplies when they work together via our real-time data pipeline, enabling longitudinal RWE capture and early signal detection.
On the scalability front, everything is cloud-native with autoscaling capable of supporting both nimble 50-patient pilots and 10,000-patient global Phase III trials. We’ve validated this through multi-region deployments across therapeutic areas with consistent system performance and uptime.
Regulatory alignment is not an overlay; it’s embedded. Every module is built with native support for 21 CFR Part 11, GDPR, HIPAA, and ICH-GCP. Features like e-signatures, data locking, version control, and immutable logs come pre-integrated. And with HealthNode, our blockchain infrastructure, compliance becomes dynamic, every transaction, edit, or consent update is automatically recorded and auditable.
Let me share a real example: A sponsor needed to scale a Phase II oncology trial from 5 to 25 sites across 8 countries after early efficacy results. With a traditional system, this would’ve taken months. With our modular suite, we scaled and reconfigured the trial infrastructure in under three weeks without disrupting ongoing workflows or retraining site staff.
One of the most valuable outcomes of this architecture is innovation velocity. When we update Hakase’s AI capabilities or enhance HealthNode’s cryptographic logic, those improvements propagate across all modules instantly no need for platform-wide updates or revalidations. This allows us to evolve continuously without compromising quality or compliance.
And flexibility isn’t just technical, it’s commercial. Sponsors can start small and scale as needed. CROs can white-label selected modules. Academic researchers can pick only what fits their study design. That universality is what’s made the suite adaptable across biotech, pharma, and academic environments alike.
Full-Spectrum AI Enablement: Bringing Clarity to the Clinical Lifecycle
[Dex]: The adoption of AI across the clinical development continuum has introduced both efficiencies and interpretability challenges. In what ways is AKT Health, Inc. deploying AI technologies to create end-to-end visibility and consistency across phases of clinical trial design, execution, monitoring, and reporting?
[Dr. Aditya]: The interpretability challenge is exactly why we built Hakase AI with “transparent intelligence.” Every recommendation comes with three things: confidence score, reasoning pathway, and alternative scenarios. When we suggest protocol changes, you see the complete evidence chain, not just the answer.
But here’s our breakthrough: “regulatory-ready AI.” Every Hakase decision is automatically documented with full audit trails. When regulators ask “Why did you modify this parameter?” – we show the complete chain of evidence, not just the recommendation.
The consistency piece is crucial. Traditional operations use different AI for recruitment, safety, analysis, each interpreting data differently. Hakase maintains “contextual memory” across the entire lifecycle. The same engine that optimized your protocol is monitoring safety and generating regulatory reports.
Result? 35% faster regulatory submissions with 100% acceptance rates because the logic is transparent and evidence trails are bulletproof.
Human-Centric Design: Elevating Patient and Site Experience through Technology
[Dex]: Modern clinical trials require digital solutions that prioritize usability without compromising scientific rigor. How are you integrating human-centric design principles into AKT Health’s technology stack to improve engagement, reduce dropout rates, and enhance the overall patient and site experience?
[Dr. Aditya]: We started with a simple premise: every data point is a person. So technology should feel like support, not surveillance.
Take our Pulse platform – we’ve built multilingual eConsent with AI comprehension scoring. Patients can pause, ask questions, or replay sections in their native language. The system adapts to their literacy level and cultural context. The result? 90% eConsent completion rates versus industry averages of 60-70%.
Our Impakt Health SaMD focuses on motivational psychology. Instead of clinical forms, patients see progress dashboards, achievement unlocks, and personalized insights about their health journey. We’re not gamifying for fun – we’re using behavioral science to sustain engagement. That’s why we maintain 85% retention rates.
For Impakt Life wearables, we chose passive capture over active engagement. Our smart ring and ECG patches collect HRV, temperature, and biomarkers without requiring any patient action. No charging reminders, no daily tasks – just seamless data flowing into Hakase AI for analysis.
Site teams get a different kind of human-centric design through Datakapt. Coordinators see protocol timelines as visual maps, not text documents. Visit prep is automated – when a patient is scheduled, the system pre-loads relevant forms, flags potential issues, and suggests conversation topics based on recent app activity.
Here’s our design philosophy: if a workflow adds cognitive burden, we either automate it or eliminate it. We measure every interaction against mental load scores. That’s not just good UX – it’s better science, because stressed users make more errors.
The proof is in retention: our platforms consistently deliver 30-40% lower dropout rates than traditional trials. When technology feels supportive rather than intrusive, people engage differently with research.
When technology truly serves human needs – when it reduces burden rather than adding it, when it feels intuitive rather than intimidating, when it enhances rather than replaces human connection – adoption follows naturally. Patients engage more deeply, sites operate more efficiently, and ultimately, we generate better data and better outcomes.
That’s not just good design philosophy – it’s good science. Because at the end of the day, clinical trials are fundamentally about people helping people. Our job is to make sure technology amplifies that human connection rather than getting in the way of it.
Living Ecosystems: Dynamic Operations for Continuous Learning
[Dex]: Traditional static models in clinical operations often fail to adapt to real-time challenges. What does it mean to reimagine clinical operations as a living, learning ecosystem, and how is AKT Health applying this paradigm to create more adaptive, resilient trial infrastructures?
[Dr. Aditya]: This is probably the concept I’m most excited about, because it represents a fundamental shift in how we think about clinical operations. Traditional models are static, the protocol is fixed, processes are linear, and decisions often lag behind real-world change. But clinical environments are anything but static. Patients evolve, contexts shift, and data flows continuously.
A living ecosystem approach means building clinical operations that can sense, learn, and adapt in real-time while maintaining scientific integrity and regulatory compliance. Think of it like the difference between a traditional garden with fixed planted rows versus a permaculture system that adapts to changing conditions while maintaining overall health and productivity.
At AKT Health, we’ve embraced what we call Ecosystem Intelligence, the idea that our platforms should function like a living organism. Every interaction within the Impakt Clinical Suite, from eConsent via Pulse to continuous RPM through Impakt Life, becomes part of a dynamic, interconnected feedback system.
Here’s an example: If multiple sites show patient confusion on eConsent, Hakase suggests content edits within IRB-approved bounds. If vitals in a region trend downward during a heatwave, Hakase reduces visit requirements or switches to virtual consults all while staying in compliance.
It’s a continuous loop: Observe → Interpret → Act → Evolve. We’ve already prevented deviations, flagged safety risks days earlier, and rescued enrollment timelines by adapting mid-study.
This isn’t theory, it’s how we make trials resilient, adaptive, and human-centered.
Strategic Horizon: The Future of AKT Health Under Your Leadership
[Dex]: The future of clinical development will be shaped by the convergence of data, platforms, and patient expectations. Looking ahead, what is your vision for AKT Health, Inc. over the next 3–5 years, particularly in redefining digital health partnerships, clinical trial transformation, and innovation leadership?
[Dr. Aditya]: The next chapter is about orchestration, not just innovation. When I look toward the future, I see us at the cusp of what I call the “Clinical Intelligence Revolution” – a fundamental transformation in how clinical research gets conducted, similar to how the internet revolutionized communication or how smartphones changed computing.
AKT Health has established itself as a next-generation life sciences platform transforming clinical, medical, and commercial functions through intelligent, technology-first solutions. Over the next three to five years, our vision is to scale this foundation globally while deepening our capabilities as the intelligence infrastructure of clinical research.
Through AKTClinical, our clinical innovation engine, we’re already redefining global trials through fully decentralized and hybrid models powered by AI and blockchain. The Impakt Clinical Suite, featuring Datakapt for eSource and ePRO, Pulse for site engagement, Impakt Health as our PMDA-registered SaMD, and Impakt Life‘s medical-grade wearables is delivering up to 90% faster execution with superior data quality and global scalability.
The next chapter focuses on expanding this ecosystem. Hakase AI will evolve into an even more sophisticated engine, processing real-world data across therapeutic areas while maintaining seamless integration with clinical data management, CDISC compliance, and SAS programming. Our HealthNode blockchain infrastructure will become the global standard for clinical data integrity and regulatory compliance.
We’re positioning AKT Health to bridge healthcare innovation with regulatory excellence at unprecedented scale. As we expand across Asia, Europe, and the Americas, our focus remains on enabling sponsors to run smarter, faster, and truly global trials, from protocol to submission, protocol to market.
The vision is clear: make AKT Health the platform where data-driven decisions accelerate access, improve outcomes, and fundamentally shape the future of life sciences. We’re not just making clinical research faster and smarter, we’re making it more patient-centric and globally accessible.
Engr. Dex Marco Tiu Guibelondo, B.Sc. Pharm, R.Ph., B.Sc. CpE
Editor-in-Chief, PharmaFEATURES
Join Proventa International’s Clinical Operations and Clinical Trial Supply Chain Europe Strategy Meeting at Crowne Plaza London Docklands, London, United Kingdom on the 9th of October 2025 to learn more about AKT Health.

Subscribe
to get our
LATEST NEWS
Related Posts

Interviews
Financing the Future of Health: Navigating Biotech Investment Risks and Rewards with David Crean, Cardiff Advisory LLC & 1004 Venture Partners
A deep dive into how strategic biotech investments balance scientific innovation with financial foresight in an era redefining the economics of health.

Interviews
The Economics of Innovation: Optimizing Clinical Trial Investments with Robert Gabriel, Ashur Capital
A thought-provoking discussion on aligning scientific rigor, venture strategy, and financial intelligence to maximize R&D efficiency and accelerate the path from discovery to market.

Interviews
Redefining Evidence Generation: Integrating eCOA Science into Biotech Development with Saima Khakwani, Clario
Saima Khakwani shares how biotechs can accelerate development and improve data quality through robust eCOA science, meaningful endpoints, and patient-centric digital measurement.

Interviews
Clinical Research 4.0: Bridging Global Standards with Local Realities and Expanding Patient Access with Rania Alshami, PDC CRO
Rania Alshami leads PDC-CRO in advancing clinical research across the Middle East and Africa by integrating global scientific standards with regional realities to accelerate patient access to innovative therapies.
Read More Articles
Spatial Collapse: Pharmacologic Degradation of PDEδ to Disrupt Oncogenic KRAS Membrane Localization
PDEδ degradation disrupts KRAS membrane localization to collapse oncogenic signaling through spatial pharmacology rather than direct enzymatic inhibition.
Neumedics’ Integrated Innovation Model: Dr. Mark Nelson on Translating Drug Discovery into API Synthesis
Dr. Mark Nelson of Neumedics outlines how integrating medicinal chemistry with scalable API synthesis from the earliest design stages defines the next evolution of pharmaceutical development.
Zentalis Pharmaceuticals’ Clinical Strategy Architecture: Dr. Stalder on Data Foresight and Oncology Execution
Dr. Joseph Stalder of Zentalis Pharmaceuticals examines how predictive data integration and disciplined program governance are redefining the future of late-stage oncology development.
Exelixis Clinical Bioanalysis Leadership, Translational DMPK Craft, and the Kirkovsky Playbook
Senior Director Dr. Leo Kirkovsky brings a rare cross-modality perspective—spanning physical organic chemistry, clinical assay leadership, and ADC bioanalysis—to show how ADME mastery becomes the decision engine that turns complex drug systems into scalable oncology development programs.
Policy Ignition: How Institutional Experiments Become Durable Global Evidence for Pharmaceutical Access
Global pharmaceutical access improves when IP, payment, and real-world evidence systems are engineered as interoperable feedback loops rather than isolated reforms.







