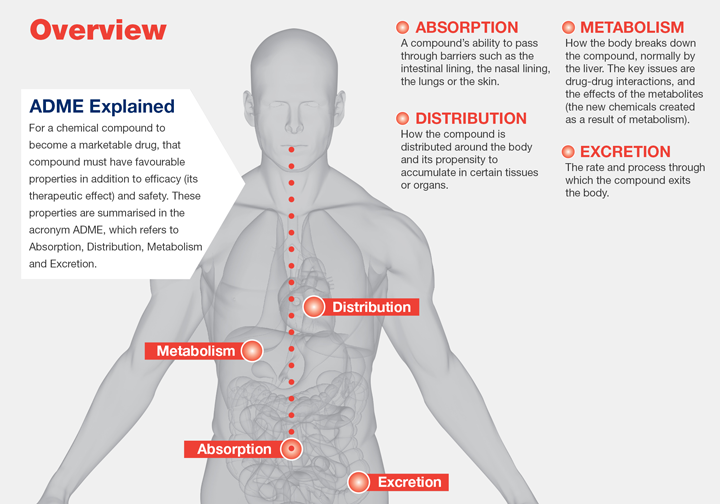
Bioavailability is a fundamental concept in pharmacology that delves into the intricate dynamics of drug absorption and distribution within the body. It encapsulates the extent and rate at which a drug reaches its intended site of action or systemic circulation. This critical measure plays a pivotal role in determining the efficacy and safety of pharmacotherapeutic interventions.
Impact of Route of Administration and Dosage
The route of administration (ROA) and dosage of a drug wield significant influence over its bioavailability. A delicate balance exists wherein the dose of a drug inversely correlates with its bioavailability. Altering the ROA necessitates adjustments in dosage to achieve optimal therapeutic outcomes. For instance, oral administration subjects drugs to gastrointestinal absorption and hepatic first-pass metabolism, potentially altering their bioavailability. In contrast, intravenous administration bypasses these processes, ensuring immediate delivery to systemic circulation and maximizing bioavailability.
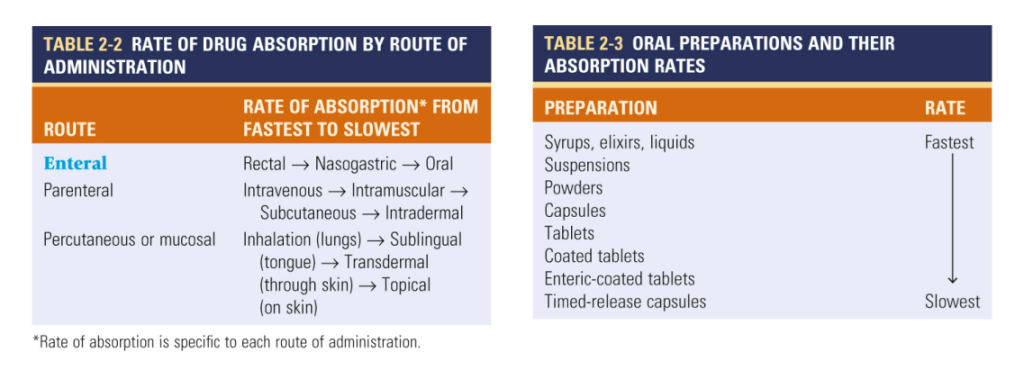
What is Drug Clearance?
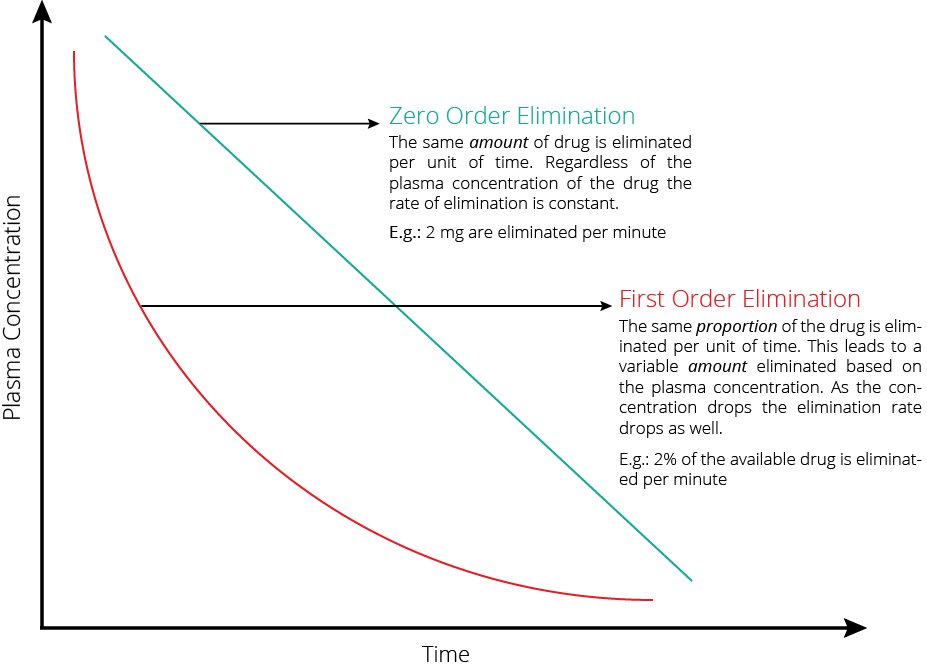
Drug clearance, the process by which active drugs are removed from systemic circulation, is a critical determinant of bioavailability. The distinction between first-order and zero-order kinetics guides our understanding of drug elimination dynamics. While first-order kinetics entail exponential elimination proportional to plasma concentration, zero-order kinetics denote constant drug elimination over time. Awareness of a drug’s clearance mechanism is paramount, as it directly impacts bioavailability and steady-state concentration, crucial considerations in therapeutic efficacy assessment.
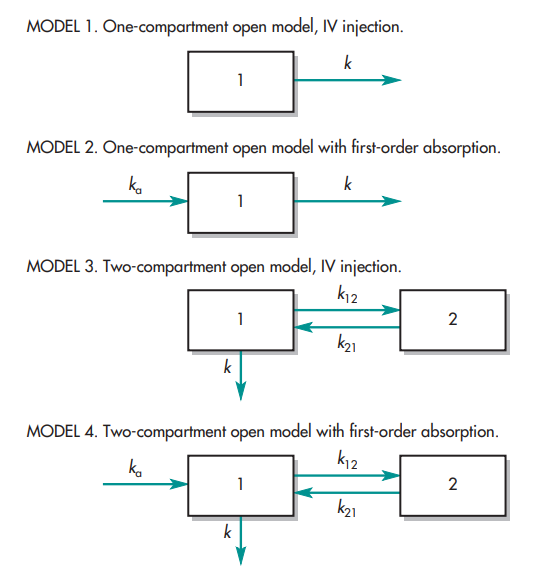
Exploring Distribution Dynamics
Distribution, the dispersion of drugs across various bodily compartments, illuminates the complexity of bioavailability. Whether in a single-compartment or multi-compartment model, the volume of distribution (Vd) serves as a key indicator of a drug’s distribution profile. A larger Vd implies extensive distribution beyond the central compartment, potentially influencing bioavailability. Understanding these distribution dynamics is instrumental in optimizing drug delivery strategies to maximize therapeutic efficacy.
Quantifying Bioavailability
Bioavailability, often quantified through area under the curve (AUC) analysis, provides invaluable insights into drug absorption kinetics. By comparing AUCs of different administration routes, one can delineate bioavailability with precision. However, inherent limitations exist, particularly concerning assumptions of constant clearance and uniform drug distribution within plasma.
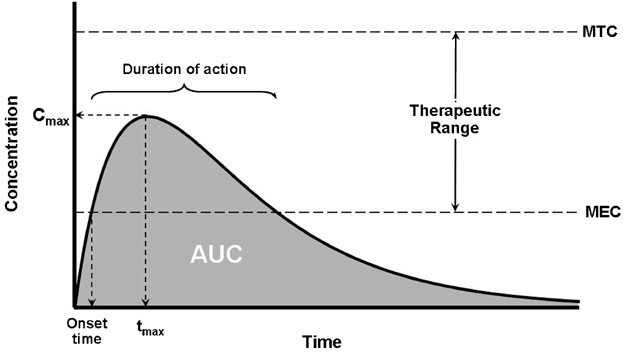
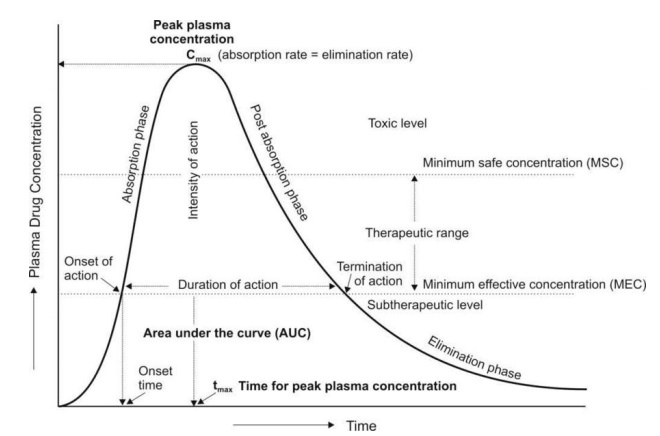
Navigating Clinical Relevance
In clinical practice, appreciation of bioavailability nuances is indispensable for informed decision-making. Interactions with intrinsic and extrinsic variables, such as metabolic pathways and concurrent medications, can significantly alter bioavailability. These considerations are exemplified in scenarios involving drugs like warfarin and nitroglycerin, where bioavailability discrepancies dictate dosing strategies tailored to individual patient needs.
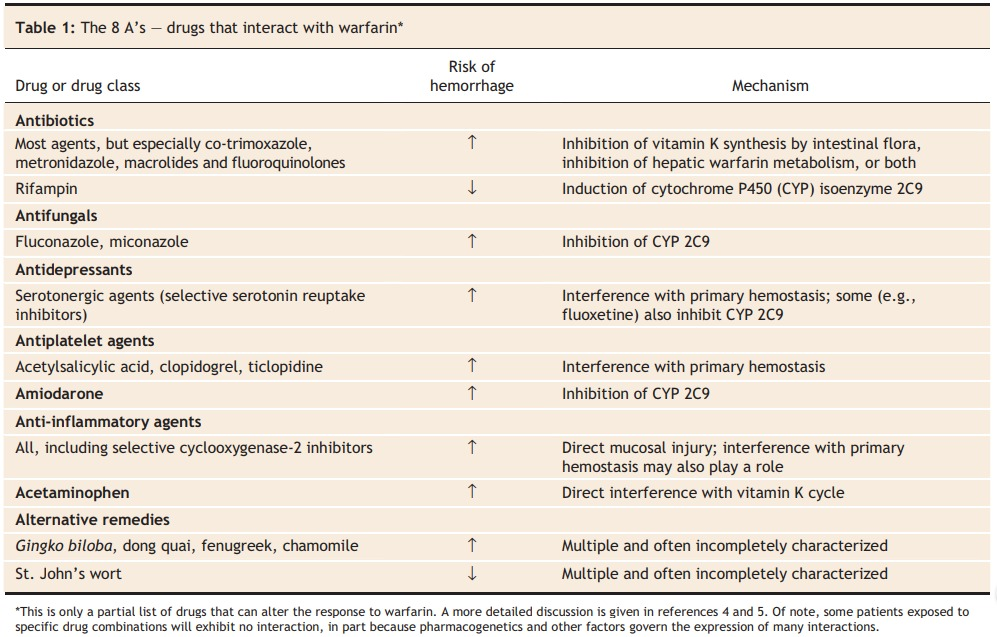

Optimizing Therapeutic Strategies
Utilizing bioavailability data empowers clinicians to devise tailored pharmacotherapeutic regimens. Equations facilitating dose calculation based on bioavailability, dosage, and clearance offer a systematic approach to dosage determination. Whether prescribing loading doses for treatment initiation or maintenance doses for long-term therapy, understanding bioavailability is paramount in achieving desired clinical outcomes while mitigating adverse effects.
Empowering Precision Medicine
In the intricate realm of pharmacology, bioavailability emerges as a linchpin guiding therapeutic precision. Its multifaceted interplay with drug administration, clearance, and distribution underscores its significance in optimizing pharmacotherapeutic interventions. By unraveling the mysteries of bioavailability, clinicians can navigate the complex landscape of drug efficacy and safety, ushering in a new era of precision medicine tailored to individual patient needs.
Engr. Dex Marco Tiu Guibelondo, B.Sc. Pharm, R.Ph., B.Sc. CpE
Editor-in-Chief, PharmaFEATURES

Subscribe
to get our
LATEST NEWS
Related Posts

Medicinal Chemistry & Pharmacology
Igor Nasonkin and Phythera Therapeutics: Moving Oncology Beyond Single Targets into Engineered Polypharmacologic Systems
Igor Nasonkin’s systems-driven approach at Phythera Therapeutics reframes oncology drug development from single-target inhibition to AI-enabled polypharmacologic network modulation using nature-derived molecular architectures.

Medicinal Chemistry & Pharmacology
Drugging mRNA with Small Molecules: David Weitz on Platform Strategy at Syrna Therapeutics
David Weitz of Syrna Therapeutics explores how small molecule modulation of mRNA, enabled by AI-driven discovery and platform-centric execution, is redefining the boundaries of druggable biology.

Medicinal Chemistry & Pharmacology
Leaner Molecules: Structural Simplification Outperforming Brute-Force Potency Potency Chasing in Lead Optimization
Structural simplification is the science of turning chemically overbuilt leads into more efficient, drug-like molecules without surrendering their therapeutic logic.
Read More Articles
Inside Johnson & Johnson’s External Innovation Engine: Devin Swanson on Translating Integrated Discovery into Strategic Value
Devin Swanson’s leadership at Johnson & Johnson Innovative Medicines redefines external innovation as a tightly governed, AI-enabled translational system integrating multi-modal drug discovery, biomarker strategy, and capital-efficient execution.
From DMPK to Distributed Execution: Mehran F. Moghaddam’s Systems Strategy at OROX BioSciences, Inc.
A systems-level examination of how Mehran F. Moghaddam operationalizes DMPK, externalized R&D, and lipid-mediated therapeutics into a predictive, high-velocity biotech development architecture.
From Data to Decision: Shicheng Guo’s Systems Approach to AI-Enabled Drug Development
A systems-level analysis of how Shicheng Guo is architecting AI-driven, human data–centric drug development at Arrowhead Pharmaceuticals.